 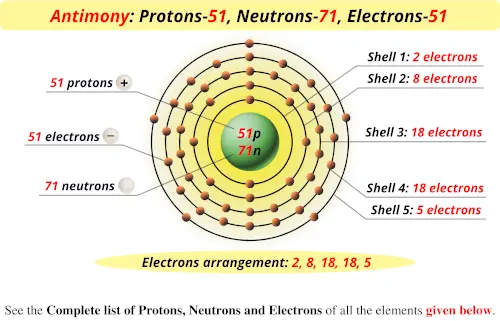
Antimony Electronegativity according to Paulingĭetermination of electronegativity of the element becomes significant to determine the bond polarity between the bond-forming atoms. The atomic weight of antimony is 121.76 amu (atomic mass unit). Antimony Atomic WeightĪtomic weight is determined by the total number of protons and neutrons along with consideration of the nuclear binding energy. Let us check the atomic numbers of Sb.Īntimony has an atomic number of 51 which indicates it has 51 protons in its nucleus. The atomic number is a unique property of an element because no two different elements have the same number of protons in their nucleus. Let us see the clock of antimony.Īntimony belongs to the p-block because it has a partially filled p-block (5p 3) in its electron configuration. Antimony Block in Periodic TableĪ block in the periodic table consists of a set of elements having similar atomic orbitals, valence electrons, etc. The group number of antimony is 5 in the periodic table, and it is placed between tin (Sn) and tellurium (Te). 
The atoms placed in the same period (horizontal rows in the periodic table) show an equal number of electron shells in the outer part of the nucleus. It is placed between arsenic (As) and bismuth (Bi). 
Let us explore the group of Sb.Īntimony belongs to group 15 of the periodic table. The atoms belonging to the same group (vertical column in the periodic table) possess similar valence shell electronic configurations. This short form comes from the Latin name “Stibium”. Let us search for the symbol of antimony. Antimony SymbolĪ chemical symbol is a short form or abbreviation used to write or describe a chemical element. Let us describe the group and period of antimony in the periodic table, melting as well as boiling point, isotopes, allotropes, ionization energies, and chemical classification with detailed explanations. Elemental antimony possesses a layered structure that consists of a fused, ruffled six-membered ring. It is a hard, brittle metalloid compound. Let us focus on the chemical properties of antimony.Īntimony is refined industrially from stibnite by roasting and followed by reduction in presence of carbon or direct reduction of stibnite with iron. Antimony is a gray-colored lustrous metalloid that is found mainly as Sb 2S 3. Also, it is important to note that the number of electrons is equal to the number of protons in a neutral atom. If in place of mass number, number of protons and neutrons are given, the sum of the two will give the value of mass number. For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. These different types of helium atoms have different masses (3 or 4 atomic mass units), and they are called isotopes. For instance, stable helium atoms exist which contain either one or two neutrons, but both the atoms have two protons. Īdditional Information: Atoms of the same element are known to have differing numbers of neutrons in their nucleus. On adding we get $121.856$ ( $ \sim 121.9$ ) and the unit will be $gm/mol$. Average atomic mass \[ = 123$īy putting the values in the equation, we haveĪverage atomic mass $ = (0.572 \times 121) (0.428 \times 123)$ Then we will calculate the average mass using the average atomic mass formula for isotopes. Hint:In order to find the answer we need to first convert the percentages into fractions by dividing them by \. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
